-
 In vitro stability of biosimilar insulin SAR341402 in the Medtronic MiniMed insulin pumps - Journal of Pharmaceutical Sciences
In vitro stability of biosimilar insulin SAR341402 in the Medtronic MiniMed insulin pumps - Journal of Pharmaceutical Sciences
-
 FDA Warns Medtronic Over Insulin Pumps | MedTruth - Prescription Drug & Medical Device Safety | Informed Advocacy
FDA Warns Medtronic Over Insulin Pumps | MedTruth - Prescription Drug & Medical Device Safety | Informed Advocacy
-
Guidance Industry
-
 Design and analysis high-risk medical device clinical trials for monitoring and treatment: review | Journal Pancreatology
Design and analysis high-risk medical device clinical trials for monitoring and treatment: review | Journal Pancreatology
-
 Study and Data Analysis of Artificial Pancreas Device Systems Closed-Loop Glucose-Sensing Insulin Delivery
Study and Data Analysis of Artificial Pancreas Device Systems Closed-Loop Glucose-Sensing Insulin Delivery
-
 July 2011 edition of Pharmacy Practice News by McMahon Group - Issuu
July 2011 edition of Pharmacy Practice News by McMahon Group - Issuu
-
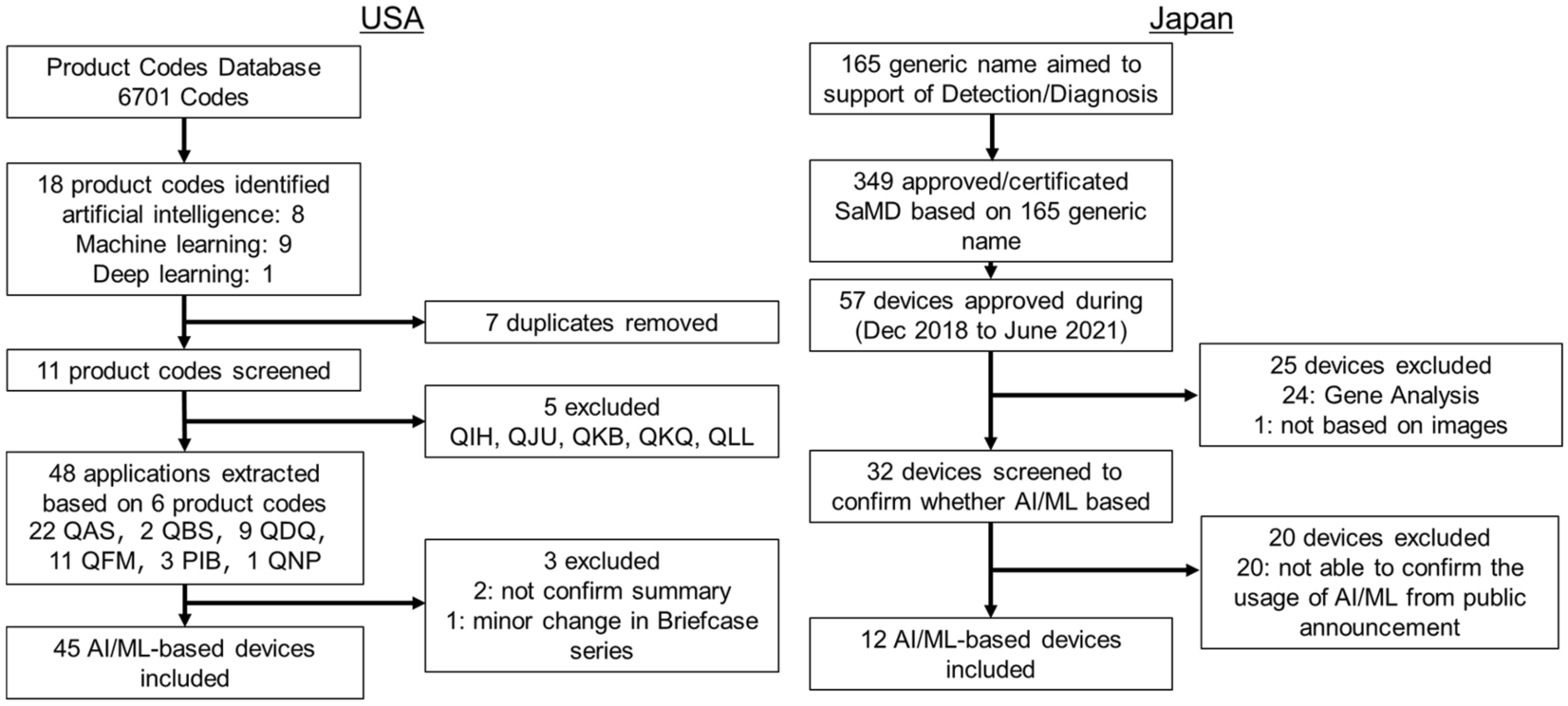 Systematic analysis of the test design and performance of AI/ML-based medical devices approved for triage/detection/diagnosis in USA Japan | Scientific
Systematic analysis of the test design and performance of AI/ML-based medical devices approved for triage/detection/diagnosis in USA Japan | Scientific
-
 An FDA Viewpoint Unique for Medical-Device Clinical | NEJM
An FDA Viewpoint Unique for Medical-Device Clinical | NEJM
-
 The Food and Drug Administration's (FDA's) 510(k) Process: A Systematic Review of 1000 - The American of Medicine
The Food and Drug Administration's (FDA's) 510(k) Process: A Systematic Review of 1000 - The American of Medicine
-
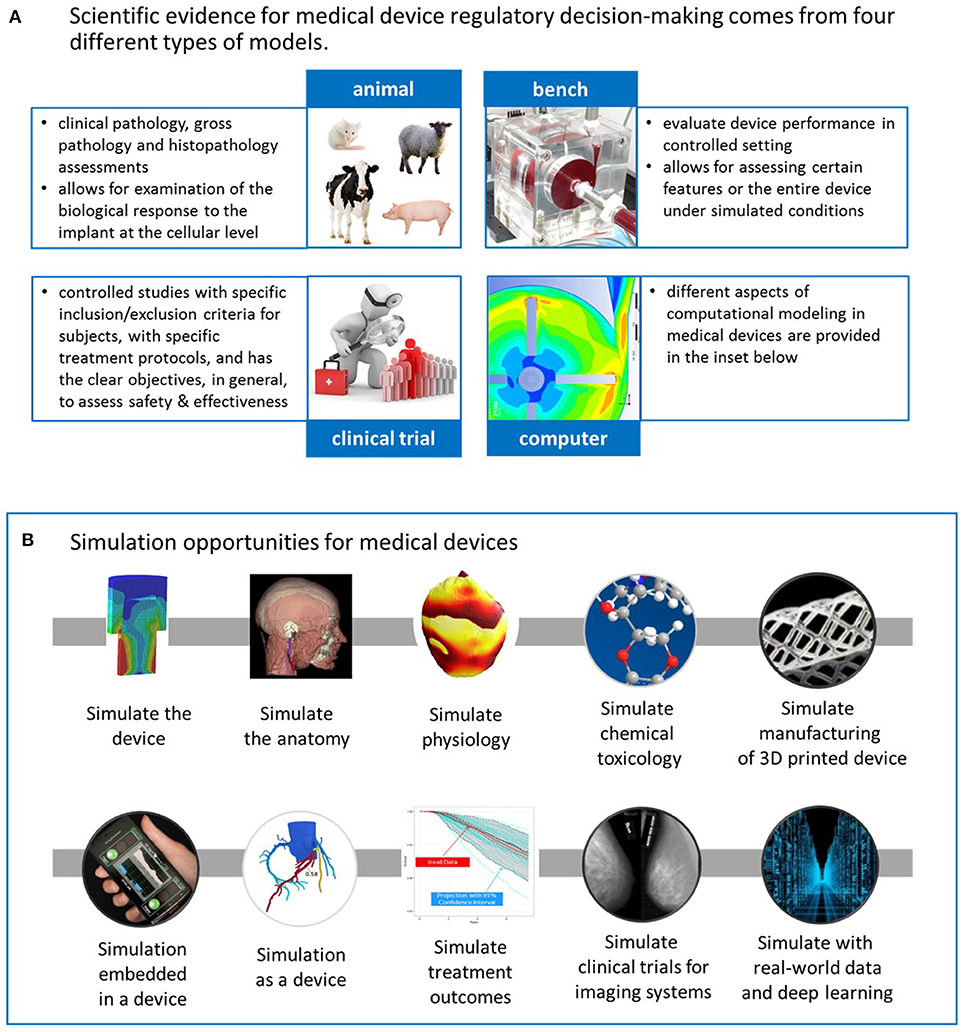 | Advancing Regulatory Science With Modeling for Medical Devices at FDA's Office of Science and Engineering
| Advancing Regulatory Science With Modeling for Medical Devices at FDA's Office of Science and Engineering
-
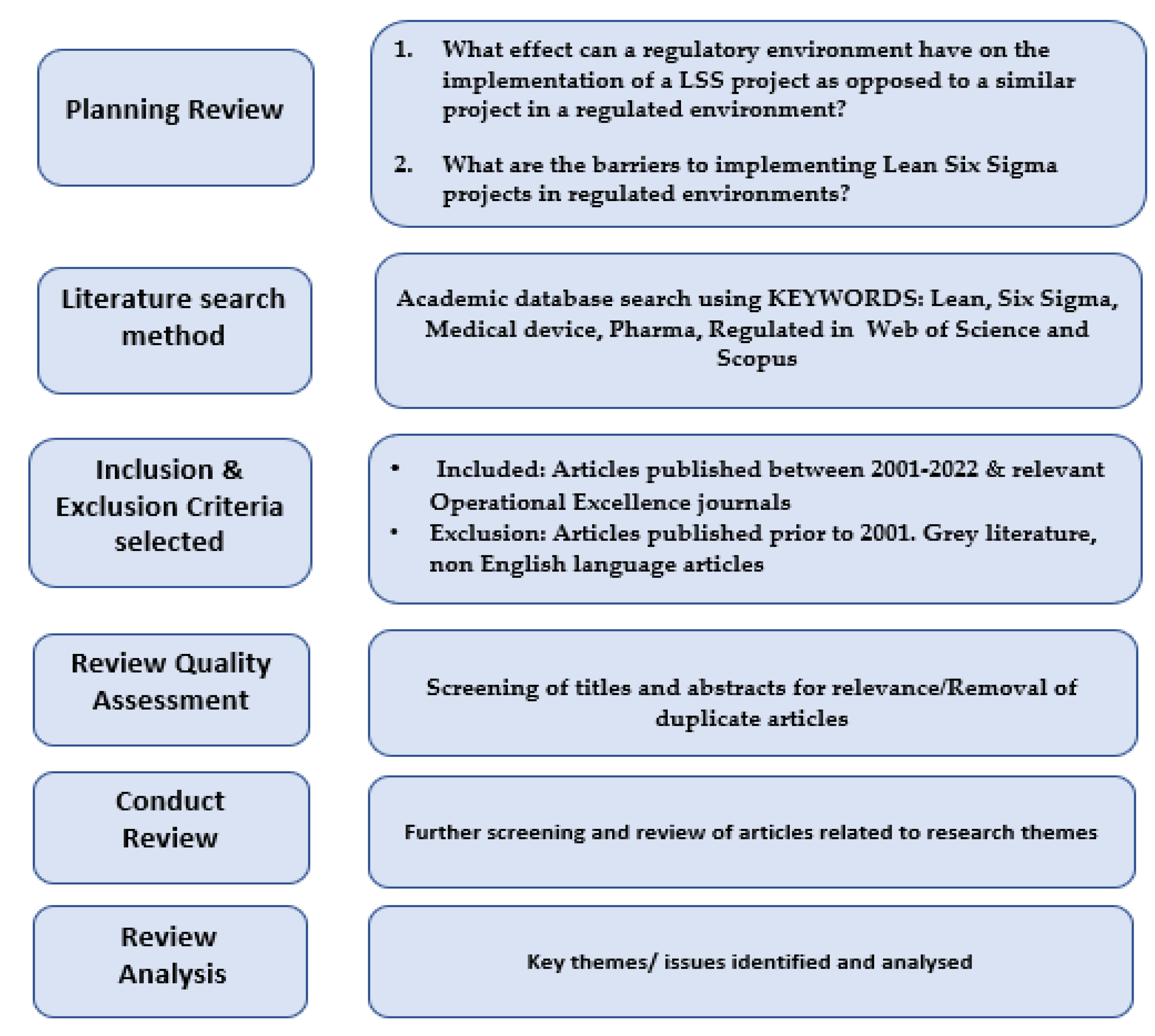 Processes | Free Full-Text | The Effect of Medical Device Regulations on Deploying a Lean Six Sigma
Processes | Free Full-Text | The Effect of Medical Device Regulations on Deploying a Lean Six Sigma
-
 Utility of the FlowMet™ Peripheral Blood Flow Monitoring System Extremity Revascularization - Today
Utility of the FlowMet™ Peripheral Blood Flow Monitoring System Extremity Revascularization - Today
-
1 §
-
 Long-Term Clinical Effectiveness of a Drug-Coated Balloon for the Treatment Femoropopliteal Lesions | Circulation: Cardiovascular Interventions
Long-Term Clinical Effectiveness of a Drug-Coated Balloon for the Treatment Femoropopliteal Lesions | Circulation: Cardiovascular Interventions
-
Hearing and Devices Including Wearable, Bone Anchored and Semi-Implantable – Individual Policy
-
 How Regulates Intelligence in Products The Pew Charitable Trusts
How Regulates Intelligence in Products The Pew Charitable Trusts
-
 New Medical Device and Therapeutic Approvals in State of the Art Review of 2021 - Alexander Choi, Michael J. Brenner, Gorelik, Isaac D. Erbele, Matthew G. Crowson, Prajoy
New Medical Device and Therapeutic Approvals in State of the Art Review of 2021 - Alexander Choi, Michael J. Brenner, Gorelik, Isaac D. Erbele, Matthew G. Crowson, Prajoy
-
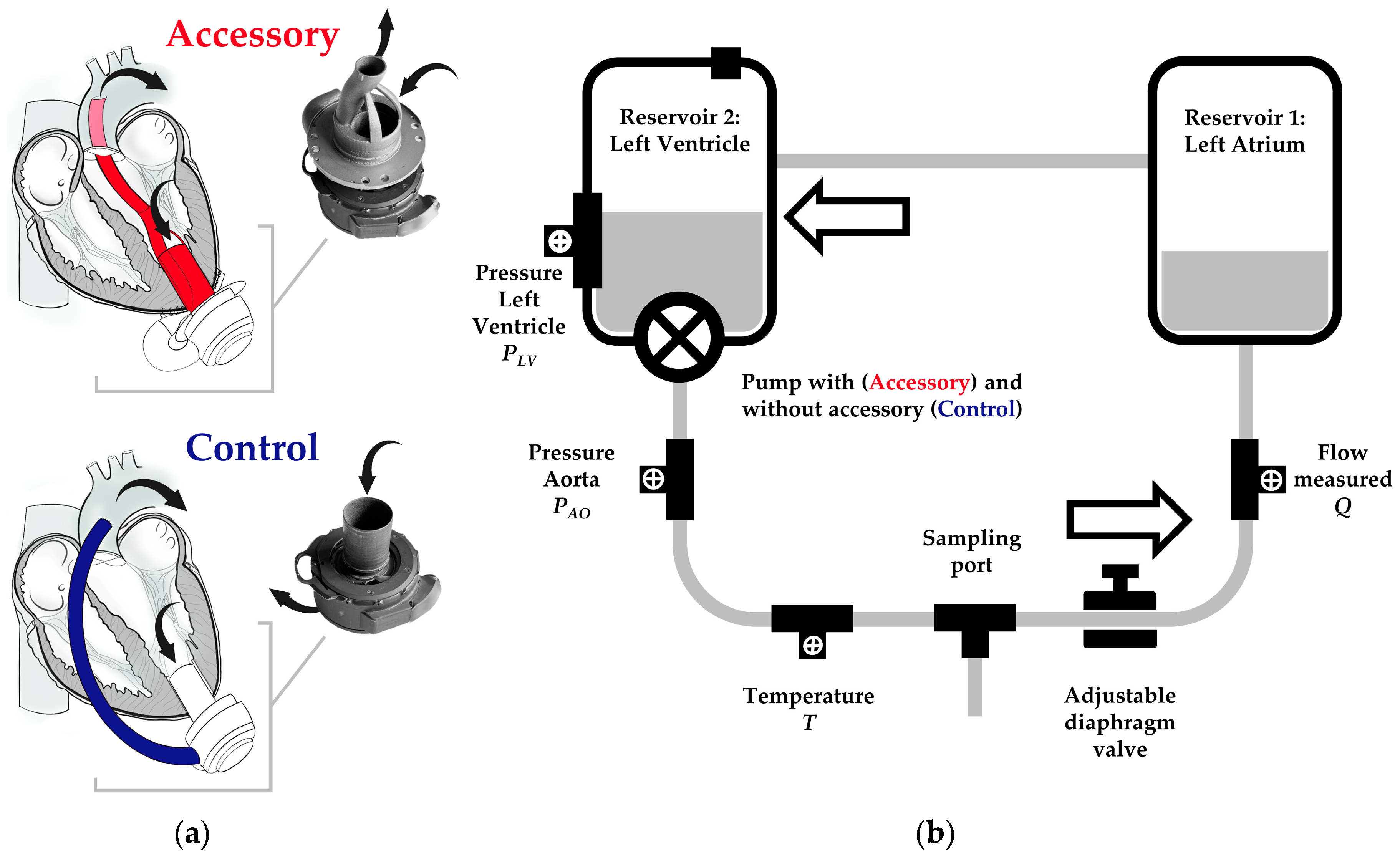 Bioengineering | Free Full-Text | of an Accessory for Left Ventricular Assist Devices on Device Flow and Pressure Head In Vitro
Bioengineering | Free Full-Text | of an Accessory for Left Ventricular Assist Devices on Device Flow and Pressure Head In Vitro
-
Certain Medtronic MiniMed Insulin Have Potential Cybersecurity Risks: FDA Safety Communication
-
 years of insulin: celebrating past, present and future of therapy | Nature Medicine
years of insulin: celebrating past, present and future of therapy | Nature Medicine
-
OF AND EFFECTIVENESS DATA (SSED)
-
 TAG® Conformable Thoracic Stent Graft with System | Gore Medical
TAG® Conformable Thoracic Stent Graft with System | Gore Medical
-
510(k) Summary of Safety and
-
 Flash Glucose Monitoring: A Patient's and Clinician's Caveats and Concerns - Practice
Flash Glucose Monitoring: A Patient's and Clinician's Caveats and Concerns - Practice
-
 CV RA Template Version - Accessdata Fda - Sign Printable Template
CV RA Template Version - Accessdata Fda - Sign Printable Template